Datenschutzerklärung
Wissenschaftliche Forschung am Institut für Veterinär-Epidemiologie und Biometrie der Freien Universität Berlin - Datenschutzerklärung
Die Möglichkeit, zu Zwecken der Wissenschaft Daten zu erheben, ist ein wertvolles Gut. Wir sind uns der großen Verantwortung bewusst, dieses Gut zu schützen und das Vertrauen zu honorieren, das uns durch Teilnehmende im Bereich von Befragungen und Messdatenerhebungen entgegengebracht wird.
Im Sinne einer transparenten Wissenschaft und um die Vorgaben des Datenschutzgesetzes zu erfüllen, geben wir allen Interessierten im Folgenden einen Überblick, welche Ziele wir mit der Erhebung von Daten für die Wissenschaft an unserem Institut grundsätzlich verfolgen und welche Maßnahmen zum Schützen aller erhobenen Daten ergriffen werden.
Daten unterstehen berechtigterweise dem gesetzlichen Schutz. Die Erhebung wissenschaftlicher Daten sowie ihre Auswertung (Verarbeitung) erfolgt an unserem Institut mit der notwendigen Sorgfalt gemäß den Vorgaben der geltenden datenschutzrechtlichen Gesetzgebung und der guten wissenschaftlichen Praxis (IVEB - Gute wissenschaftliche Praxis).
Die Mitwirkung von Dritten an wissenschaftlichen Projekten ist zu jeder Zeit freiwillig und erfolgt stets nur nach Aufklärung über Ziel und Art der Datenerhebung und informierter Einwilligung zur Teilnahme. Grundsätzlich werden alle Teilnehmenden vor Studienbeginn in geeigneter Weise über den Zweck der jeweiligen Datenerhebung, deren Verarbeitung und ggf. der geplanten Weitergabe in leicht nachvollziehbarer Form informiert und stimmen der Teilnahme an der Studie ausdrücklich zu.
Allen Teilnehmenden an vergangenen wie künftigen Projekten möchten wir an dieser Stelle für die Unterstützung danken.
1. Warum ist es notwendig, dass Daten zu wissenschaftlichen Zwecken erhoben werden?
Angesichts vieler ungeklärter Fragen in Bezug auf Vorbeugung und Therapie zahlreicher Krankheiten sind wissenschaftliche Erkenntnisse für die Weiterentwicklung der Gesundheitsversorgung von Haus- und Nutztieren unersetzbar. In der Wissenschaft dienen die im Rahmen von Studien erhobenen Daten unterschiedlicher Art dazu, zuverlässige Antworten auf eine spezifische Forschungsfrage (z.B. Wie viele Hunde haben Diabetes und welche Probleme treten bei der Behandlung auf?) zu finden.
Nur auf der Grundlage geeigneter Daten können Wissenslücken geschlossen und passende Lösungen für Probleme erarbeitet werden (schematisch dargestellt in Abbildung 1).Dabei werden die notwendigen Daten in der Regel entweder im Rahmen einer Befragung oder einer Messung ermittelt. Die so gewonnenen Fakten werden in Datenbanken zusammengeführt, strukturiert und mit Hilfe von statistischen Verfahren ausgewertet, um allgemeingültige Aussagen treffen und basierend auf diesem Wissenszusatz Lösungsvorschläge oder Handlungsempfehlungen formulieren zu können.
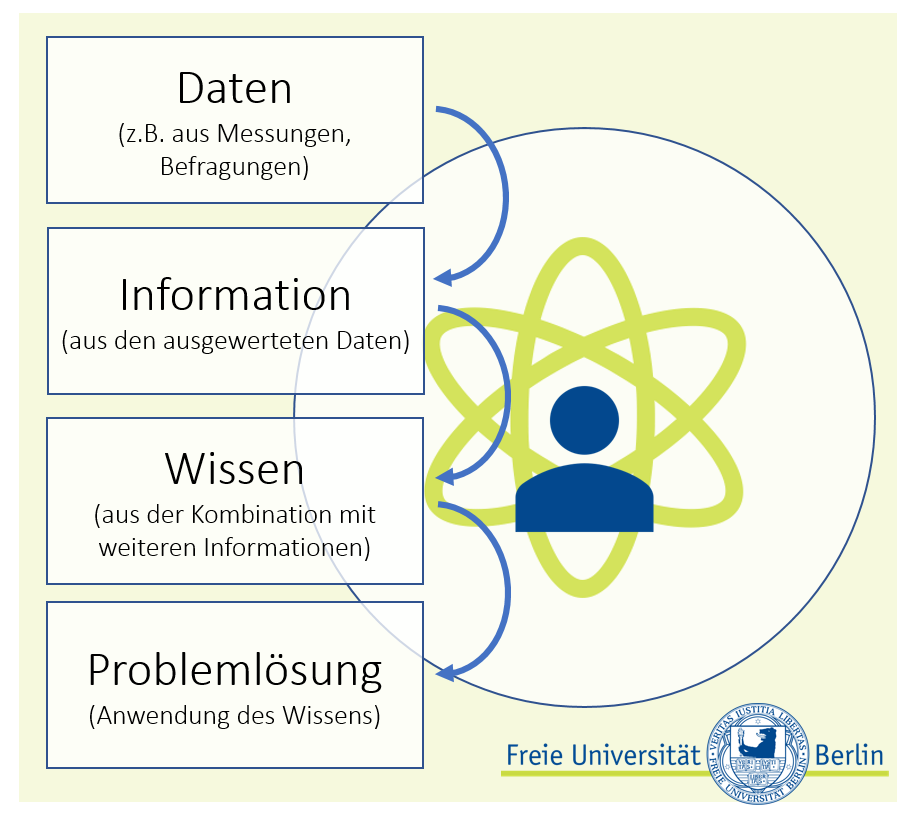
Abbildung1: Daten in der Wissenschaft
2. Warum werden im Rahmen von Befragungen häufig Daten wie Geschlecht, Alter und Einkommen abgefragt?
In der Regel können die Daten nicht an der gesamten Population erhoben werden (z.B. können nicht alle Hundehalter in Deutschland befragt werden), sondern es wird eine Stichprobe befragt. Um im Anschluss feststellen zu können, ob die Stichprobe ein gutes Abbild der Gesamtpopulation liefert, müssen entsprechende demographische Daten wie das Geschlecht und das Geburtsjahr mit erhoben werden. Zeigen die demographischen Daten, dass die Stichprobe der Gesamtpopulation ähnlich ist, kann die Erhebung als „repräsentativ“ betrachtet werden. Die demographischen Daten dienen also primär der Bewertung der Stichprobenqualität, auch wenn sie mit der eigentlichen Fragestellung nichts zu tun haben. Bei demographischen Daten handelt es sich nicht um personenbezogene Daten, auch wenn die Fragen im ersten Moment ggf. persönlich erscheinen. Zum Unterschied zwischen „personenbezogen“ und „persönlich“ siehe auch Punkt 6.
3. Sind anhand der Daten Rückschlüsse auf meine Person möglich?
Alle am Institut für Veterinär-Epidemiologie und Biometrie erfassten Daten werden lediglich für wissenschaftlich-statistische Zwecke erhoben. Die Erhebung erfolgt wann immer möglich anonymisiert. Die Identität einer teilnehmenden Person kann mit rechtlich zulässigen Mitteln nicht bestimmt werden. Sollten im Rahmen spezifischer Fragestellungen Rückschlüsse auf die Identität einer befragten Person potentiell möglich sein (z.B. im Rahmen eines persönlichen Interviews), so wird dieser Personenbezug nicht erfasst oder durch Pseudonymisierung hinfällig gemacht. Die für die Fragestellung relevanten Daten werden also nicht personenbezogen, sondern anonymisiert oder pseudonymisiert gespeichert und weiter ausgewertet.
4. Was passiert mit den Daten?
Schritt 1: Erhebung und Speicherung
Die Erhebung von wissenschaftlichen Daten kann auf unterschiedlichen Wegen erfolgen:
Im Rahmen von Umfragen werden mit Hilfe von online und/oder offline verfügbaren Fragebögen Antworten von meist zahlreichen Teilnehmenden (= quantitative Forschung) zu spezifischen Fragestellungen erfasst. Gemäß den Anforderungen an gute wissenschaftliche Praxis werden ausschließlich solche Fragen gestellt, die zur Erfüllung des Forschungszwecks erforderlich und angemessen sind. Entsprechend werden personenbezogene Daten hier nur in Ausnahmefällen und gemäß den Vorgaben der DSGVO nur nach eindeutiger informierter Einwilligung durch die Teilnehmenden erhoben.
Im Rahmen von persönlichen Interviews oder bei der Erfassung von Daten im Rahmen klinischer oder epidemiologischer Studien (z.B. Probennahmen in Milchviehbetrieben) kann es notwendig werden, personenbezogene Daten zu erheben. So muss beispielsweise die Adresse bekannt sein, um einen Studienbetrieb anfahren zu können, und Interviewpartner stellen sich einander mit Namen vor. In diesen Fällen werden personenbezogene Daten zwar erhoben, vor der Auswertung jedoch aus dem Datensatz entfernt und nicht für die Auswertung genutzt.
Für die Untersuchung biologischer Proben und die Nutzung der Untersuchungsergebnisse gelten gesonderte Vorschriften, die den gleichen Grundsätzen des Datenschutzes unterworfen sind. In Studien, in denen biologische Proben entnommen und untersucht werden, werden die Teilnehmer gesondert über die vorgesehenen Untersuchungen aufgeklärt und stimmen der Untersuchung und ggf. der weiteren Verwendung der entnommenen Proben ausdrücklich zu.
Alle erhobenen Daten werden auf Servern der Freien Universität gespeichert und obliegen den entsprechenden hohen Schutzbestimmungen. Der Zugriff durch unberechtigte Personen ist unter Einsatz legaler Mittel zu keiner Zeit möglich.
Im Rahmen von wissenschaftlichen Forschungsprojekten erhobene Daten müssen grundsätzlich für die Dauer von zehn Jahren gespeichert werden, um im Zweifelsfalle Überprüfungen durch daraus gewonnenen Erkenntnisse durchführen zu können.
Schritt 2: Verarbeitung/Auswertung
Die statistische Auswertung der Daten erfolgt stets nicht-personenbezogen und dient dem Aufdecken der zugrundeliegenden relevanten Beziehungen (z.B. dem Zusammenhang zwischen der Fütterung und dem Auftreten von Diabetes beim Hund). In Fällen der Erhebung personenbezogener Daten werden diese im weiteren Verlauf der Auswertung anonymisiert oder pseudonymisiert. Die statistische Auswertung erfolgt mit Rechnern der Freien Universität Berlin.
Schritt 3: Veröffentlichung
Die Veröffentlichung der Daten erfolgt lediglich in zusammengefasster, kondensierter Form. Rohdaten werden grundsätzlich nicht veröffentlicht.
Schritt 4: Datennutzung und -weitergabe
Eine Weitergabe von Daten an Einrichtungen außerhalb des Instituts erfolgt normalerweise nicht. Eine Ausnahme stellt die weitere Verwendung der Daten für wissenschaftliche Methoden dar, für die Daten aus mehreren Studien zusammengeführt werden müssen. Diese sogenannte Sekundärdatennutzung ist ebenfalls gesetzlich geregelt und erfolgt anhand geeigneter technischer und organisatorischer Methoden, die eine missbräuchliche Verwendung der Daten verhindern.
Die Rohdaten der wissenschaftlichen Erhebung einschließlich möglicher personenbezogener Daten werden nicht oder nur nach ausdrücklicher informierter Einwilligung an Dritte weitergegeben. Im Rahmen von Auftragsforschungsprojekten/externen Finanzierungen werden keine Rohdaten an die Auftraggebenden weitergegeben, um Manipulationen oder eine unrechtmäßige weitere Verarbeitung zu verhindern.
Die erhobenen Daten werden zu jeder Zeit während und nach Erhebung und Auswertung vertraulich behandelt und ausschließlich zu wissenschaftlichen Zwecken verwendet. Dennoch können internetbasierte Datenspeicherung und -übertragung grundsätzlich Sicherheitslücken aufweisen, sodass ein absoluter Schutz nicht gewährleistet werden kann.
5. Wer ist mein Ansprechpartner in Fragen des Datenschutzes?
In erster Instanz liegt die Verantwortung für die Einhaltung des Datenschutzes im Rahmen von Forschungsprojekten bei der Projektleitung, in zweiter Instanz bei der Institutsleitung.
Die Freie Universität Berlin hat eine Datenschutzbeauftragte/einen Datenschutzbeauftragten, die/der bei Datenschutzfragen und für Datenschutz-Folgeabschätzungen (Artikel 35 DS-GVO) beratend zur Verfügung steht.
6. Was bedeutet eigentlich…
Personenbezogene Daten?
Personenbezogene Daten sind Informationen, mit Hilfe derer direkte oder indirekte Rückschlüsse auf eine bestimmte natürliche Person erfolgen können, mit denen also eine Person eindeutig identifiziert und mit einem Datensatz in Verbindung gebracht werden kann. Dies können beispielsweise ein Name, eine Kennnummer, Standortdaten, eine Online-Kennung oder andere besondere Merkmale sein, die Ausdruck der physischen, physiologischen, genetischen, psychischen, wirtschaftlichen, kulturellen oder sozialen Identität dieser natürlichen Person sind.
Zu unterscheiden sind diese personenbezogenen Daten von Daten, die zwar persönlich sind (bspw. die häufig gestellten Fragen nachdem Bildungsabschlussoder der Einkommensgruppe), die aber keine direkten oder indirekten Rückschlüsse auf eine bestimmte Person erlauben und lediglich einer demographischen Vergleichbarkeit der Stichprobe dienen.
Verarbeitung?
Verarbeitung ist jeder Vorgang oder eine Reihe von Vorgängen, der/die mit oder ohne Hilfe automatisierter Verfahren im Zusammenhang mit Daten ausgeführt wird. Dazu gehört das Erheben, das Erfassen, die Organisation, das Ordnen, die Speicherung, die Anpassung oder Veränderung, das Auslesen, das Abfragen, die Verwendung, die Offenlegung durch Übermittlung, Verbreitung oder eine andere Form der Bereitstellung, der Abgleich oder die Verknüpfung, die Einschränkung, das Löschen oder die Vernichtung von Daten.
Anonymisierung und Pseudonymisierung?
Unter Anonymisierung versteht man ein Verfahren, bei dem die Zuordnung von Daten zu einer bestimmten natürlichen Person nicht möglich ist und auch nicht wieder hergestellt werden kann.
Bei der Pseudonymisierung hingegen können die Daten einer bestimmten natürlichen Person zugeordnet werden. Die notwendigen (personenbezogenen) Daten werden jedoch von den übrigen Daten getrennt aufbewahrt und sind lediglich über einen Identifier (z.B. eine Codenummer) miteinander verknüpft. Zur Sicherung des Datenschutzes hat eine Person immer nur Zugang entweder zu den personenbezogenen Daten oder zu den Studiendaten. Pseudonymisierung ist zur Sicherstellung einer repräsentativen Stichprobe notwendig, wenn z.B. in zwei Regionen gleich viele Personen teilnehmen sollen.
Zur besseren Anschaulichkeit ist derUnterschied zwischen anonymisierten und pseudonymisierten Daten in Abbildung 2 grafisch dargestellt.
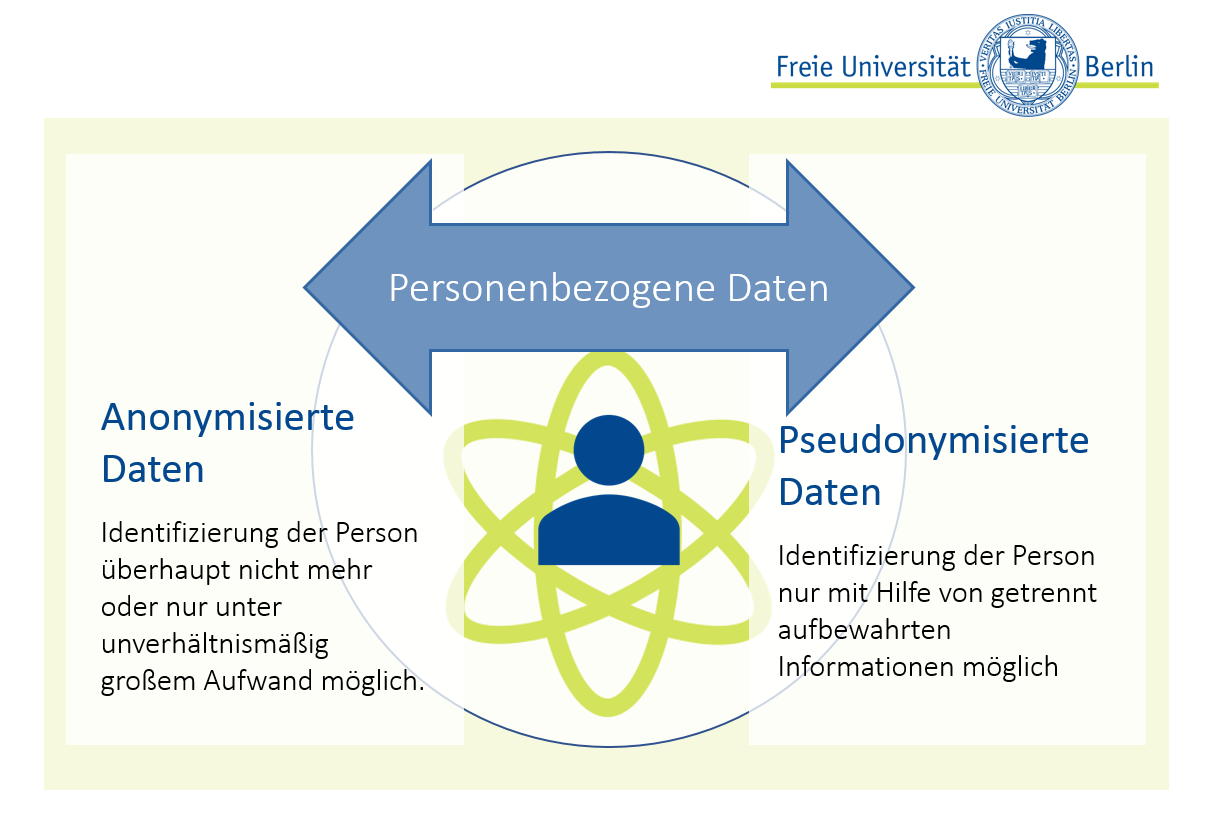
Abbildung 2: Anonymisierte und pseudonymisierte Daten
Einwilligung?
Eine Einwilligung ist die unmissverständlich abgegebene Willensbekundung in Form einer Erklärung oder einer sonstigen eindeutigen bestätigenden Handlung (z.B. aktives Setzen eines Hakens), mit der eine betroffene Person zu verstehen gibt, dass sie mit der Verarbeitung der sie betreffenden personenbezogenen Daten einverstanden ist.Die Einwilligung muss von der betroffenen Person freiwillig für jeden Fall in informierter Weise getroffen werden.
Recht auf Löschen/Vergessenwerden?
Gemäß Art. 17 der DS-GVO hat jede/r Teilnehmende das Recht, dass Daten, die im Zusammenhang mit personenbezogenen Daten erhoben wurden, gelöscht werden. Ebenfalls besteht das Recht, dass Daten nachträglich berichtigt werden können (Art. 13 Abs. 2 lit. B DS-GVO). Zur Durchsetzung dieses Anspruchs genügt der formlose schriftliche oder mündliche Widerruf an die jeweilige Projektleitung oder die Institutsleitung.